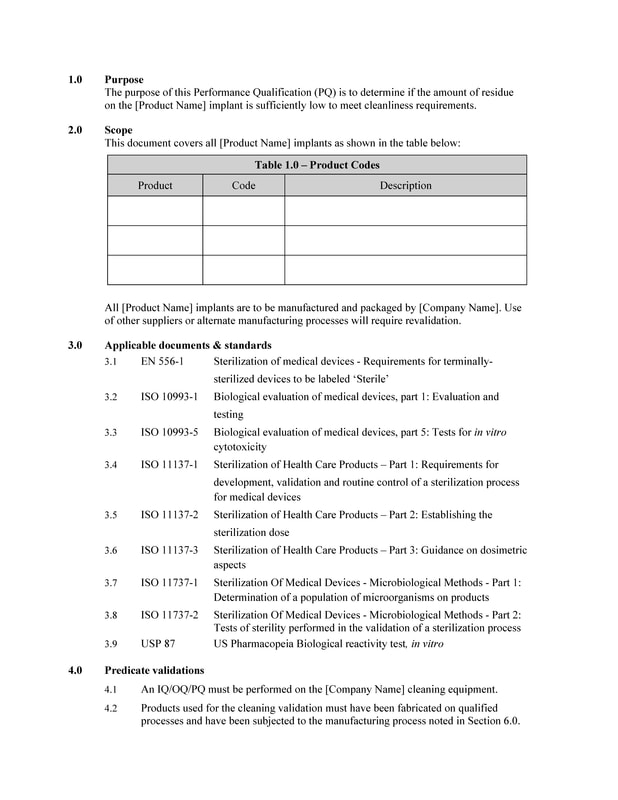
- STORE
- >
- VALIDATION PROTOCOLS
- >
- Final Cleaning Orthopedic Implants
Final Cleaning Orthopedic Implants
SKU:
PQ-FC
$149.00
$149.00
Unavailable
per item
The final cleaning performance qualification (PQ) captures the cleanliness requirements of orthopedic implants and can be applied to implants made of CoCrMo, Polyethylene, PEEK, Titanium and/or Titanium and Ha coated implants. The protocol includes the test method and strategy, sample size requirements, and acceptance criteria for TOC, THC and Cytotoxicity testing.
- FDA Compliant
- Adaptable to Multiple Cleaning Systems
- Adaptable to Multiple Materials
- MS Word Format
- Digital Product - Instant Download
Final Cleaning Orthopedic ImplantsThe final cleaning performance qualification (PQ) protocol captures the cleanliness requirements of orthopedic implants and can be applied to implants made of CoCrMo, Polyethylene, PEEK, Titanium and/or Titanium and Ha coated implants.
The protocol includes the test method and strategy, sample size requirements, and acceptance criteria for Bioburden, TOC, THC and Cytotoxicity testing. For more information about our implant final cleaning PQ protocol contact us at info@aplyon.com |
|
Customer's also viewed
Biocompatibility - Toxicity Testing Procedure | ISO 13485 | FDA QSR Compliant
$199.00
The Biocompatibility Procedure governs the biological evaluation of materials and medical devices. The Biocompatibility Procedure is aligned with ISO 10993-1, 2018, ISO 14971:2019, MDR 2017/745 and FDA QSR.
- ISO 10993-1, 2018 Compliant
- ISO 14971, 2019 Compliant
- ISO 13485:2016 Compliant
- FDA QSR Compliant
- MDR 2017/745 Compliant
- MS Word Format
- Digital Content - Instant Download
Risk Management Procedures Bundle | ISO 13485 | FDA QSR Compliant
$499.00
The Risk Management Procedure Bundle includes four (4) ISO 14971:2019 compliant standard operating procedures relating to the risk management process for medical devices.
- EN ISO 14971:2019 Compliant
- ISO 13485:2016 Compliant
- FDA QSR Compliant
- MDR EU 2017/745 Compliant
- Includes Related Forms
- MS Word and Excel Format
- Digital Content - Instant Download
Design Control Procedure Bundle | ISO 13485 | FDA QSR Compliant
$499.00
The ISO 13485 Design Control Procedure Bundle includes design control and related procedures that adhere to the design control requirements of ISO 13485:2016 and FDA QSR.
- ISO 13485:2016 Compliant
- FDA QSR Compliant
- Includes All Related Forms
- MS Word Format
- Digital Content - Instant Download